Ultra – durable Polyurea – The material of the Present & Future (The Ultimate protective solution of the 21st Century)
- Overview
Each year, countries around the world spend hundreds of billions of dollars on repairs, maintenance, and mitigating the consequences of corrosion. Global corrosion-related losses are estimated at approximately USD 2.5 trillion annually, equivalent to 3.4% of the global GDP [NACE Report, 2016]. Metal corrosion has long been considered the “invisible enemy” of industry, particularly in critical sectors such as the petroleum industry.
In the petroleum industry, corrosion not only corrosion not only leads to product losses but can also result in oil spills, fires, explosions, and severe environmental impacts.
Recognizing the seriousness of the issue, engineers and scientists have continuously researched and improved corrosion protection solutions. From early manual methods such as oil-based paints and bitumen coatings to more advanced technologies like epoxy coatings and cathodic protection, the industry has undergone nearly a century of development.
In recent years, polyurea-based coatings – an advanced protective material – have emerged as an optimal solution due to their superior properties: rapid curing time, high mechanical strength, excellent chemical resistance, and significantly longer service life compared to traditional coating systems. With these advantages, polyurea is projected to become a dominant trend in corrosion protection technology within the petroleum industry, both now and the near future.
- Corrosion in the Petroleum industry
Metal corrosion in the petroleum industry refers to the degradation of metal surfaces in equipment, pipelines, storage tanks, offshore platforms, and other infrastructure due to the harsh conditions of exploration, production, transportation, and storage of petroleum resources. It is one of the most common and severe failure mechanisms, causing billions of dollars in losses annually, while posing significant risks to operational safety and the environment.
2.1. CO₂ Corrosion
CO₂ corrosion has long been recognized as a prevalent issue in the production and transportation of oil and gas, as carbon dioxide is one of the primary corrosive agents in these systems.
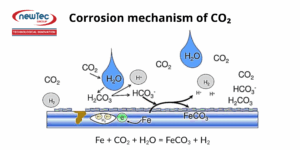
In its dry gaseous form, CO₂ does not cause corrosion at typical production temperatures. However, when dissolved in water, it forms carbonic acid, which acidifies the solution and thereby promotes electrochemical reactions between the steel and the aqueous phase in contact.
The severity of CO₂ corrosion depends on multiple factors, including temperature, pH, water chemistry, presence of a non-aqueous phase, flow conditions, and the metallurgical properties of the material. This type of corrosion is also the most common form encountered in the petroleum industry.
At elevated temperatures, a layer of iron carbide scale may form on the pipeline surface, acting as a protective barrier, but the metal can still undergo corrosion beneath this layer.
CO₂ corrosion typically manifests in two forms:
- Pitting corrosion: localized and aggressive attack that creates deep pits on the metal surface.
- Mesa attack: localized corrosion occurring under moderate flow conditions.
The corrosion mechanisms associated with CO₂ involve carbonic acid or bicarbonate ions generated when CO₂ dissolves in water.

(a) General corrosion; (b) Pitting corrosion; (c) Mesa attack; (d) Flow-induced localized corrosion
2.2 H₂S Corrosion
H₂S corrosion refers to the degradation of metal caused by exposure to hydrogen sulfide (H₂S) and moisture. It is one of the most severe forms of corrosion, causing significant damage.
Although H₂S itself is not a strong corrosive agent, in the presence of water, it becomes a considerable corrosive species and also induces sulfide stress cracking in pipelines.
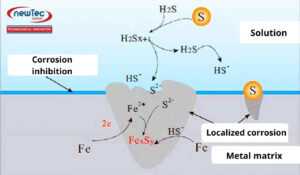
When dissolved in water, hydrogen sulfide (H₂S) forms a weak acid, releasing hydrogen ions (H⁺), which leads to corrosion processes.
Typical corrosion products include:
- Iron sulfide (FeSₓ): Forms a scale on the surface. At low temperatures, this scale can act as a temporary protective layer, slowing down the corrosion process.
- Hydrogen gas (H₂)
Common forms of H₂S-induced corrosion:
- Uniform corrosion
- Pitting corrosion
- Stepwise cracking

Corrosion due to H2S
2.3 Crevice Corrosion
Crevice corrosion is a type of localized corrosion that typically occurs in narrow gaps, crevices, or confined spaces on the metal surface, where liquid becomes trapped and cannot circulate freely.
This phenomenon arises due to concentration differences of corrosive agents on the metal surface. The electrochemical potential difference between regions leads to concentrated corrosion at the crevices or the formation of pits (pitting corrosion).
Dissolved oxygen in the drilling fluid promotes both pitting and crevice corrosion in shaded or shielded areas of the drill string. This is a common cause of washout (surface erosion or cavitation) and damage to the underlying metal beneath protective rubber coatings on the tubing.

2.4. Erosion-Corrosion
Erosion-corrosion is a phenomenon in which the corrosion rate of a metal increases due to the continuous removal of the protective passive film from the pipe surface.
This passive film is a thin, naturally formed corrosion product layer that helps stabilize and slow down the corrosion process. However, under turbulent flow and high shear stress within the pipeline, this protective layer is disrupted, leading to a rapid increase in corrosion rate.
Erosion-corrosion typically occurs in areas with turbulent, high-velocity flow and significant mechanical impacts. The severity of this phenomenon depends on flow velocity, the concentration and shape of suspended solid particles in the fluid.
High flow rates, the presence of abrasive suspended particles, along with corrosive agents in drilling and production fluids, significantly accelerate the degradation of the metal surface.
This form of corrosion is often overlooked or mistaken for pure mechanical wear (abrasion).
2.5. Microbiologically Influenced Corrosion (MIC)
Microbiologically influenced corrosion (MIC) is a type of corrosion driven by the metabolic activity of microorganisms, and it has gained significant attention in industrial settings. These microorganisms produce byproducts such as carbon dioxide (CO₂), hydrogen sulfide (H₂S), and various organic acids, which increase the aggressiveness of the fluid environment inside pipelines and cause damage to metal surfaces.
Bacterial colony formation is particularly active in aqueous environments with neutral pH, especially in stagnant water conditions. Numerous studies have documented a rich microbial presence in the water of oilfields, with significant populations of bacteria such as Bacillus, Pseudomonas, Micrococcus, Mycobacterium, Clostridium, and Escherichia.
Among these, Escherichia species are notable for containing the hydrogenase enzyme, which enables the utilization of molecular hydrogen and is involved in the cathodic depolarization process—thereby accelerating corrosion of steel casings and pipelines in oilfields. Additionally, polysaccharide-slime-producing bacteria such as Achromobacter, Flavobacterium, and Desulfuricans tend to adhere to each other tightly, forming large biofilms that accumulate on surfaces and contribute to serious blockages in injection wells.
MIC is often identified by the presence of black, slimy biofilm deposits or nodular growths on pipeline surfaces, usually accompanied by localized pitting corrosion beneath these deposits. These effects not only compromise the integrity of critical infrastructure but also require special attention from industry professionals to effectively prevent and manage microbial corrosion.
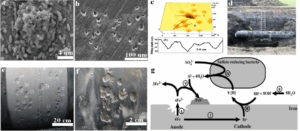
(from Metals journal (MDPI), Vol. 12, September 2022)
- (a, b) SEM images of biofilm and pitting.
- (c) Pit depth measurement using CLSM (Confocal Laser Scanning Microscopy).
- (d) Buried steel pipeline under anaerobic soil conditions; arrows indicate pitting at the weld joint (Source: [34], ©2022 Elsevier).
- (e) Weld zone with visible corrosion pits; numbers indicate pit depth in millimeters.
- (f) Magnified view of pitting at various locations along the same pipeline (Source: [22], ©2022 ASM).
- (g) Schematic of the MIC mechanism caused by sulfate-reducing bacteria (SRB) on iron surfaces (Source: [35], ©2022 MDPI).
2.6. Oxygen-Induced Corrosion (O₂)
Dissolved oxygen in water or wet gas is one of the most aggressive oxidizing agents, causing uniform electrochemical corrosion on metal surfaces.
In cooling water systems, wet gas pipelines, or unprotected equipment, oxygen ingress can lead to the formation of rust, uniform corrosion, or pitting.
Basic reactions:
Anodic: Fe → Fe²⁺ + 2e⁻
Cathodic: O₂ + 4e⁻ + 2H₂O → 4OH⁻
Overall, the presence of oxygen accelerates the oxidation of iron ions, forming hydrated iron oxides (rust), and significantly increases the rate of general corrosion.
2.7. Hydrochloric Acid (HCl) Corrosion
HCl is primarily formed from chlorine-containing compounds present in crude oil or as a byproduct of cracking processes—especially in the vapor phase when salts are present in the crude.
Basic reaction:
Fe + 2HCl → FeCl₂ + H₂↑
When HCl dissolves in condensed water, it produces a strong acid solution that aggressively corrodes carbon steel in overhead systems, storage tanks, and heat exchangers.
- Impact of corrosion on the petroleum industry
Corrosion presents a systemic challenge across the entire oil and gas value chain—from exploration and production to transportation, storage, and refining. It not only degrades equipment quality and operational efficiency but also poses major economic, safety, and environmental consequences.
Reduced equipment lifespan and reliability
Corrosion is a leading cause of rapid degradation in fixed assets such as production wells, pipelines, separators, storage tanks, distillation towers, and heat exchangers.
Wall thinning, perforations, or cracking in pipelines and vessels can lead to operational shutdowns, increased maintenance costs, and reduced equipment availability.
Significant economic burden
According to a NACE report (2002), the direct cost of corrosion in the U.S. oil and gas industry exceeded $1.3 billion per year, mainly due to maintenance and equipment replacement.
Globally, corrosion is estimated to cost about 3–4% of GDP, or roughly $2.5 trillion annually. Implementing effective corrosion management strategies could reduce these costs by 15–35%, equivalent to savings of $375 to $875 billion per year (NACE Report, 2016).
Safety and environmental risks
Corrosion is a root cause of numerous serious incidents in the oil and gas sector, including hydrocarbon, H₂S, and NH₃ leaks, leading to explosions, poisoning, and environmental pollution.
Notable incidents directly linked to corrosion-related failures include:
- Chevron Richmond Refinery (2012)
- Golden Eagle Refinery (2009)
- Prudhoe Bay pipeline (Alaska, 2006)
- Offshore gas pipeline explosion, Western Australia (2008)
- SGP Tower explosion at Humber Refinery (2001)
Beyond human casualties, businesses also suffer reputational damage and massive compensation costs.
Impact on Efficiency and Technology
Corrosion decreases heat transfer efficiency, increases energy consumption, reduces flow rates, and degrades operational performance.
Companies are compelled to invest heavily in corrosion-resistant alloys, advanced polymer coatings, environmentally friendly chemicals, and cathodic protection systems.
- Corrosion control solutions in the petroleum industry
To minimize corrosion-related losses, ensure safety, operational efficiency, and extend equipment lifespan, oil and gas facilities must implement integrated corrosion management strategies—from design and materials to operation and maintenance.
4.1. Use of appropriate corrosion-resistant materials
Select high corrosion-resistant alloys such as stainless steel, nickel-based alloys (Inconel, Monel), and titanium for aggressive environments like H₂S, HCl, and amines.
In less severe environments, carbon steel may still be used with the addition of protective coatings or cathodic protection systems.
4.2. Optimized design and operation
Design to eliminate crevices, stagnant zones, and humid hot spots prone to corrosion.
Maintain fluid velocity within recommended limits to avoid erosion-corrosion.
Control pH levels, remove salts from crude oil, and eliminate dissolved oxygen in water systems.
4.3. Use of corrosion inhibitors
Inject or blend corrosion inhibitors into gas/oil streams.
Ensure the inhibitors are environmentally friendly and low in toxicity.
4.4. Periodic inspection and monitoring
Employ advanced inspection technologies for early corrosion detection.
Monitor operational data and analyze corrosion trends.
4.5. Application of protective coatings
One of the most effective strategies is the use of polymer-based protective coatings to isolate metallic surfaces from corrosive environments. Among these, polyurea stands out as an advanced coating technology due to its exceptional properties.
- Polyurea – The material of the present and the future
Polyurea is an elastomeric polymer formed by the rapid reaction between isocyanate and polyamine, characterized by ultra-fast cure time, excellent waterproofing, high elasticity, chemical resistance, and strong adhesion to various substrates. These attributes make polyurea a promising coating solution in the oil & gas sector—where robust protection against corrosion, abrasion, and harsh environmental exposure is critical.
Applications of Polyurea in the Petroleum Industry
Corrosion and abrasion protection
The oil and gas industry constantly faces threats from corrosion and mechanical wear, which compromise the durability of assets like pump skids, storage tanks, pipelines, wellheads, drainage systems, and chemical containment areas.
Polyurea forms a durable, elastic barrier that shields these components from physical and chemical damage. Its long-term protective performance exceeds that of epoxy, polyurethane, or traditional hybrid coatings.
Waterproofing and sealing
Leaks in oil and gas infrastructure can lead to severe environmental and economic consequences. Polyurea acts as a high-performance waterproof membrane, offering rapid cure and flexibility, ideal for sealing cracks, joints, and potential leakage points.
It is applied to both interior and exterior surfaces of steel or concrete tanks, buried or aboveground tanks, pipelines, and containment areas on soil or concrete bases.
Structural reinforcement
Beyond protection, polyurea enhances structural durability and load-bearing capacity, thanks to its high elasticity and tensile strength. This helps oil and gas infrastructure withstand harsh operating conditions and reduces damage caused by mechanical loads or abrasion.
Secondary containment systems
To meet environmental protection requirements, polyurea is used in secondary containment systems to prevent chemical or oil spills from reaching the environment. Its excellent chemical resistance makes it particularly suitable for such critical applications.
Flow optimization
Polyurea coatings produce smooth, seamless surfaces, reducing fluid friction and preventing residue build-up inside pipes, fittings, or transport surfaces. This helps maintain stable flow rates, reduce maintenance frequency, and enhance operational efficiency.
Offshore protection
Harsh marine environments expose offshore structures to saltwater, chemicals, and mechanical impacts that accelerate corrosion.
Polyurea is used to protect platform decks, topside structures, mooring lines, ballast tanks, and other components from corrosion, abrasion, and impact. On drilling decks—where heavy steel pipes often drag across surfaces—polyurea has proven significantly more durable than conventional coatings.
Polyurea application for secondary containment areas
BBL Falcon Industries leveraged the surge in oil drilling activities across Texas, Oklahoma, and New Mexico by deploying polyurea spray systems for secondary containment areas around condensate storage tanks.
Instead of using traditional modular liners prone to leakage, the company applied seamless polyurea coatings over geotextile fabric and metal containment walls, ensuring superior waterproofing and durability.
BBL Falcon’s method is optimized for both new constructions and retrofitting, helping to extend service life, reduce maintenance costs, and enhance safety for oil and chemical storage zones.
Oil Sands Bitumen Tank Project, Canada

Corrosion Protection Project, Suban Trunk Lines, Indonesia

Dagang–Cangzhou Project (Northern China)
Rigid polyurea (PU) was spray-applied onto gas pipelines to provide corrosion and erosion protection under cold climate conditions.

Polyurea coating on gas pipeline installed beneath the Balonne River, Surat Basin, Queensland, Australia

Petro-Chemical Tank Farms (Alaska)

Polyurea has proven to be an advanced, efficient, and sustainable solution for corrosion protection in the petroleum industry. With rapid application capabilities, superior adhesion, excellent chemical resistance, long service life, and slow aging characteristics, polyurea significantly reduces infrastructure lifecycle costs while ensuring environmental and operational safety. In today’s industry trends, polyurea is increasingly replacing traditional coating systems and has become the preferred choice of major petroleum corporations.
- Conclusion
It can be affirmed that metal corrosion is one of the most critical challenges facing the petroleum industry—not only due to its rapid degradation of steel structures but also because of its profound economic, safety, and environmental consequences. Typical corrosion mechanisms in petroleum environments—such as CO₂ corrosion, H₂S corrosion, microbiologically influenced corrosion (MIC), and erosion-corrosion—require increasingly effective and sustainable protection solutions.
Traditional corrosion protection technologies—such as epoxy coatings, polyurethane paints, galvanizing, and PE wrapping—have played a vital role for decades. However, they have begun to show limitations under today’s harsher operational demands: shorter-than-expected service life, poor crack resistance, low impact durability, inadequate chemical and salt resistance, and extended application times, which can lead to downtime and production loss during maintenance.
With the industry’s shift toward sustainable development, VOC emission reduction, extended asset longevity, and cost optimization, polyurea-based protective coatings are expected to continue expanding their presence in the petroleum sector over the next 10 to 20 years. Currently, no alternative material offers a more complete combination of performance and durability than polyurea. Ongoing advancements in formulation and automated application processes will help establish polyurea as a new standard in industrial structure protection.
The adoption of polyurea represents more than just a change in coating material—it signifies a shift in corrosion management philosophy from passive prevention to proactive lifecycle optimization of assets and facilities. Investing in advanced technologies today is the key to ensuring safe and efficient operations for the petroleum industry in the future.
NEWTEC GROUP is proud to be a pioneer in Vietnam in successfully researching and producing high-performance polyurea coatings with quality comparable to leading industrial nations such as South Korea and Germany. We are committed to delivering durable polyurea materials and advanced application solutions to enhance asset longevity and provide sustainable value for petroleum projects at the present and in the future.





You may also be interested in
MARINE COATING SYSTEM FOR SHIPS OPERATING ACROSS SEA REGIONS
Corrosion Protection – Fouling Control – Comparison of Epoxy, PU, and Modified Polyurea In the
Mar
Newtec Group appoints Deputy General Director, enhancing management capabilities
On the morning of February 2nd, amidst the joyful and enthusiastic atmosphere of welcoming the
Mar
Newtec Group joins hands with the “Tet for the Poor – Winter for Children 2026” program in Nghe An
On January 29th, Mr. Mach Tho Cuong, representing Newtec Group Joint Stock Company, was present
Mar
WATERPROOFING AND CORROSION PROTECTION FOR THE ENERGY INDUSTRY
1. General context: As energy infrastructure faces the challenges of time. For over a century
Jan
HOT OR COLD POLYUREA?
Polyurea is becoming a strategic coating material in infrastructure, heavy industry, and civil engineering projects due to its
Dec
NEWTEC GROUP’S MARK AT VINA COATINGS 2025
At the Vina Coatings 2025 International Exhibition , Newtec Group made a strong impression with a series of groundbreaking
Dec
NEWTEC GROUP PARTICIPATED IN VINA COATINGS FAIR HO CHI MINH CITY 2025
Newtec Group is pleased to announce that it will participate in the International Paint and Coating
Nov
NEWTEC GROUP welcomes leading experts in construction materials at VIETBUILD 2025
Newtec Group is honored to welcome leading experts in the construction materials industry at the company’s
Oct